Blogs
Latest Blog Posts
Regulatory Process of In Vitro Diagnostic Medical devices in Ethiopia
Ethiopian Food and Drug Authority (EFDA) is a national regulatory body mandated to regulate food, medical products, tobaccos, and other similar products in the country before being distributed/marketed and used. Therefore, all devices which fulfil the medical device definition provided … Read More
Food and Medicine Administration Proclamation No: 1112/2019
If it is to achieve a consistent success, the primary task of any business entity should be being legally acceptable. I want you to know the legal principle- “Ignorance of the law excuses no man from practicing it”. … Read More
How to group or bundle medical devices and submit in one application to register with EFDA
From 2018 on, Food and Drug Authority (FDA) of Ethiopia started implementing the rules of appropriate bundling of different medical devices into a single registration request application. Bundling or grouping devices is a very important strategy to enable the assessors … Read More
Guidelines for “Registration Requirements of In Vitro Diagnostic Medical Devices”
In order to fully understand EFDA’s comprehensive registration requirements and make request for the Marketing Authorization of In vitro diagnostic medical devices, I recommend you to download and read the guideline that is attached below (in PDF format).
Here, I … Read More
Guidelines for “Registration Requirements of Medical Devices other than In Vitro Diagnostic Devices”
In order to fully understand EFDA’s comprehensive registration requirements and make request for the Marketing Authorization of Non In vitro diagnostic medical devices, I recommend you to download and read the guideline that is attached below (in PDF format).
Here, … Read More
How do Local Innovators of COVID-19 related Medical Products get support and approval for their products?
Since WHO declared COVID-19 as Public Health Emergency of International Concern, all countries have been putting all their efforts together to fight this pandemic.
Ethiopia, being the second most populous country in Africa and one of the most vulnerable countries … Read More
EFDA’s Interim Quick Assessment (IQA) system for Medical devices which are listed as ‘high priority devices’ due to COVID-19
The issue of CoronaVirus Disease (COVID-19) has now become beyond everything else. This is due to the disease's feature of easy spreading, no enough diagnosis, no known treatment, no enough Personal Preventive Equipment (PPE) across the world. This makes the … Read More
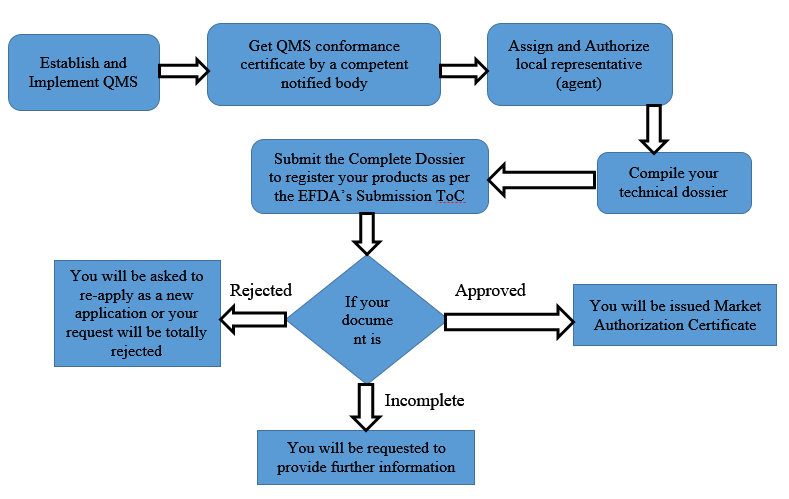
Medical Device regulation and its registration process by Ethiopian Food and Drug Authority (EFDA)
Prior to their placement on the market in the territory, Ethiopia regulates medical devices just like any other countries do even though the scope and stringency of regulation may differ. The Ethiopian Food and Drug Authority (EFDA) is mandated to … Read More
How to decide final year project topic – for Engineering Students
I have been asked a lot of questions on how to decide “new and innovative project or research topic” by so many Engineering students from different areas especially Biomedical Engineering personally and through inbox. Most of these questioners ask for … Read More